Patient support, delivered
Trial Flow enables sponsors and CROs to standardise patient support delivery across studies, sites, regions and patient populations — improving adherence, retention and trial timelines at scale.
Rapidly configured to each protocol, the Trial Flow platform gives patients and caregivers the right support at the right time, increasing their readiness to participate throughout a study.
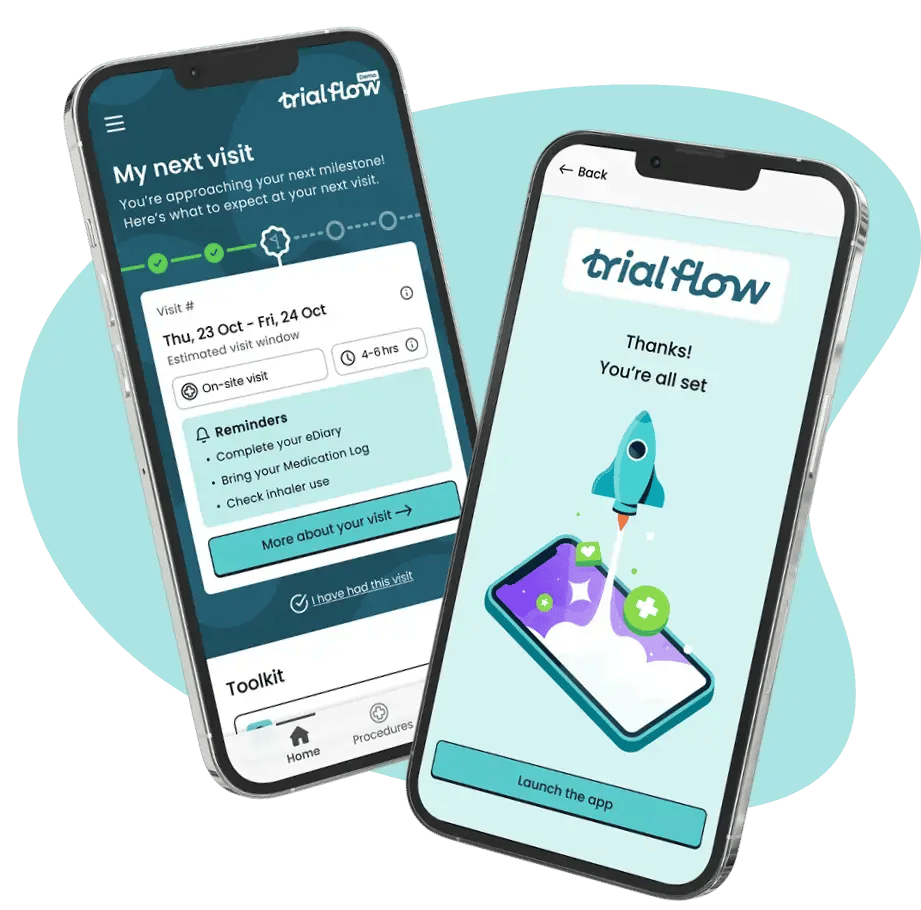
How Trial Flow works
Trial Flow turns complex protocol requirements into structured, patient-facing support that can be
deployed quickly and consistently across global studies.

Step 1: Assess
We assess each study protocol: including procedures, sites, trial visit logistics, and participant criteria to identify where patients, caregivers and site teams need support.

Step 2: Configure
We then configure Trial Flow around your study: selecting the right combination of support features, localising content by site and language, generating ethics packs and setting up compliant data collection.

Step 3: Deliver
We deliver the patient-facing study app, alongside site activation and regular reporting to support a smooth launch and provide ongoing insight into what patients engage with to drive adherence and retention.
12-16 week
deployment
Launch protocol-aligned support faster than custom app or agency-led builds.
Sponsor content integration
Make existing patient materials easier to access, track and learn from.
Over 96% schedule adherence
Our delivery model keeps trial timelines on track.
The four pillars of effective patient support
Information alone is not enough to keep participants engaged throughout a trial. Trial Flow brings together logistical, educational, distraction & coping, and motivation & progress support to address the real barriers that lead to avoidable protocol deviations, delays, and dropouts.

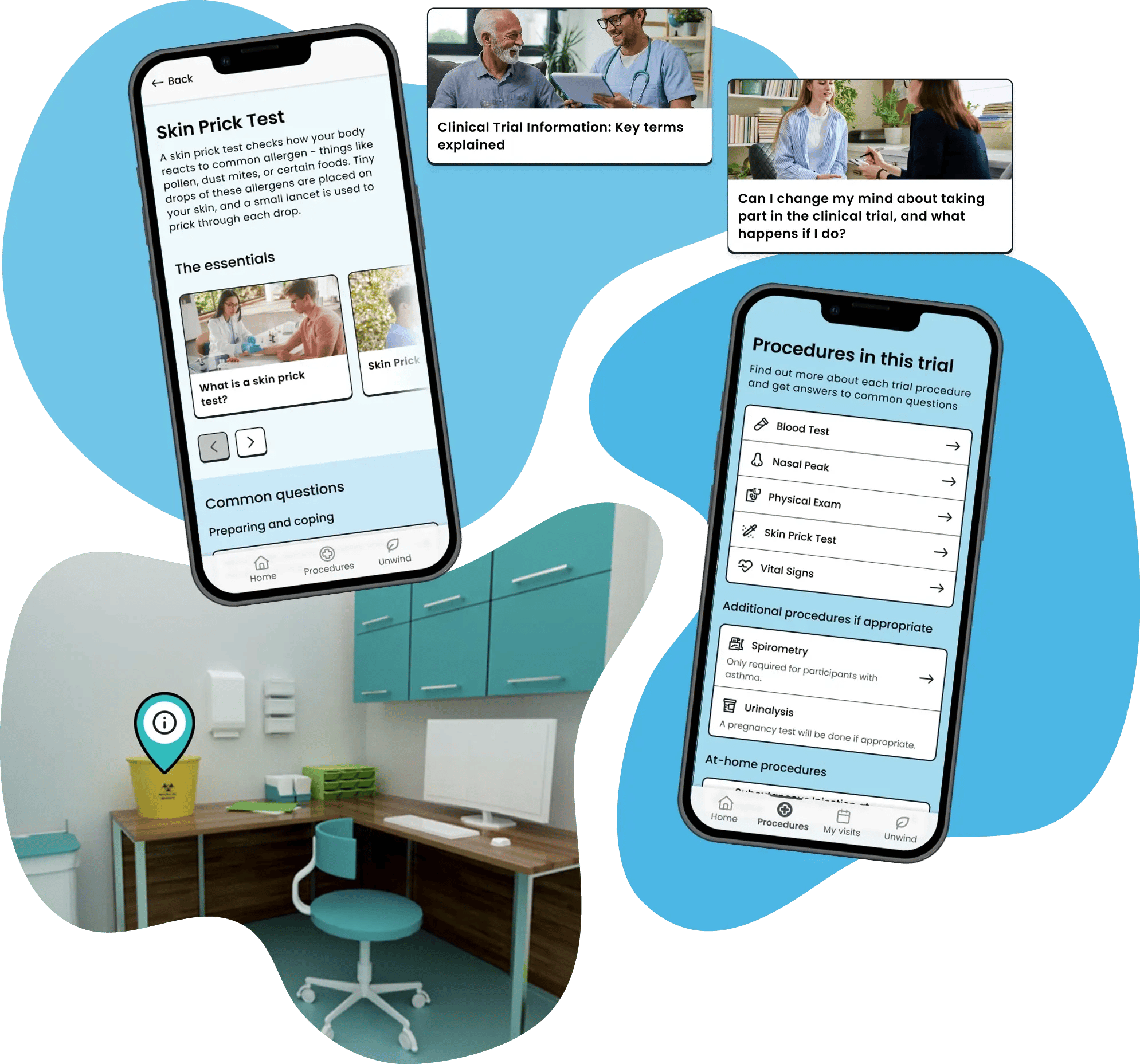
Logistical
Trial Flow's logistical support helps participants understand what is happening, when it is happening, and what they need to do next.
Feature examples:
- My Next Visit
- Notifications
- Fasting guidelines
- Notes
- Site information
Makes the practicalities of trial participation easier to navigate day-to-day
Educational
Trial Flow's educational support gives patients and caregivers clear, accessible information about different aspects of their trial, tailored to their specific protocol and user needs.
Feature examples:
- Clinical trial information
- Study information (inc. sub-studies)
- Procedure education
- Virtual tours
- Quizzes
Improves understanding of trial requirements, and the purpose behind each one, so patients can prepare and remain engaged through to study completion.
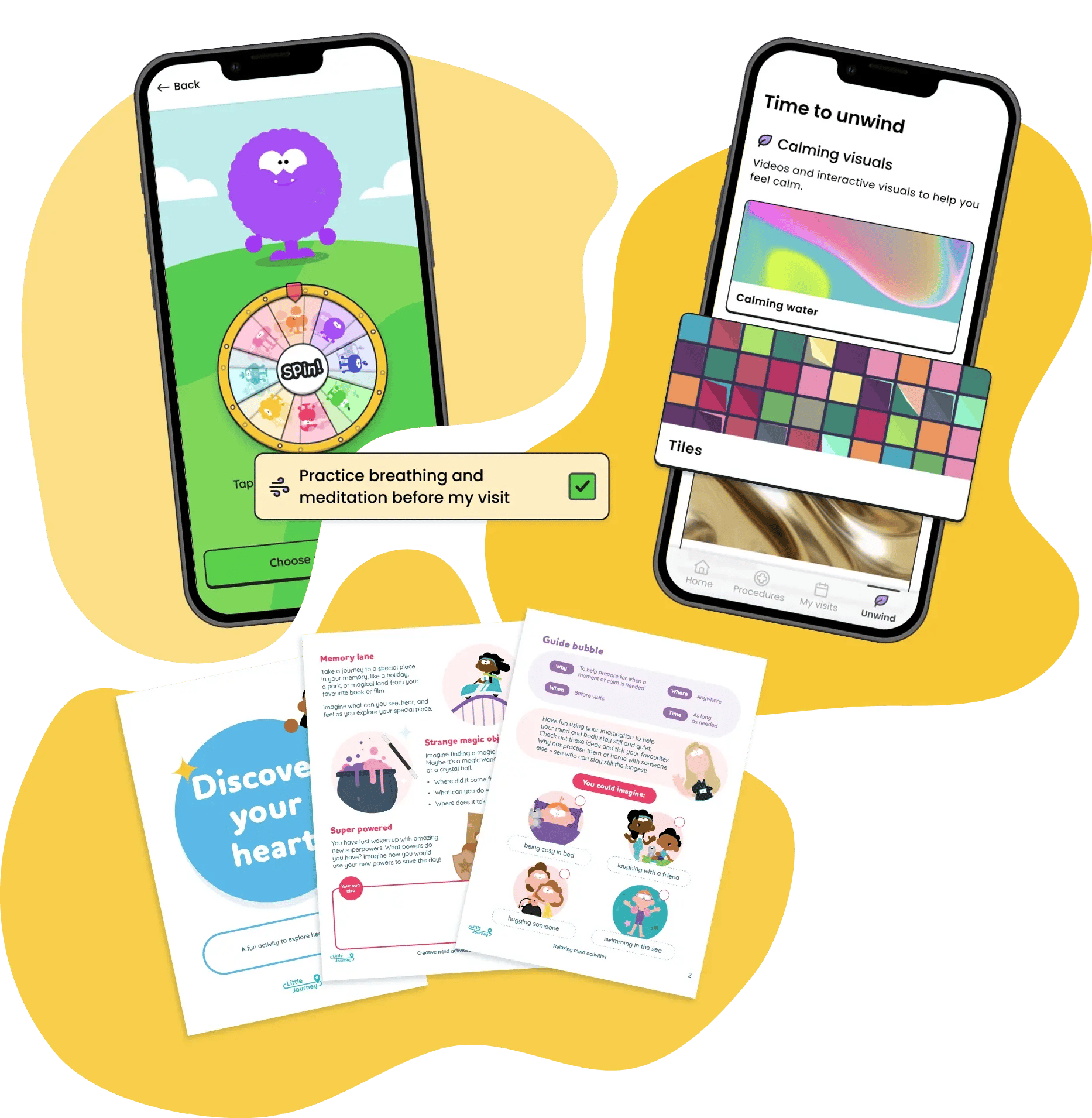
Distraction & Coping
Trial Flow's distraction & coping support helps patients manage uncertainty and cope with stressful moments throughout a trial.
Feature examples:
- My Zone (paediatrics)
- Unwind (adolescents & adults)
- Soothing activities
- Downloadable preparation activities
Reduces the emotional burden to trial participation to help patients attend visits, complete trial requirements and remain in the study.
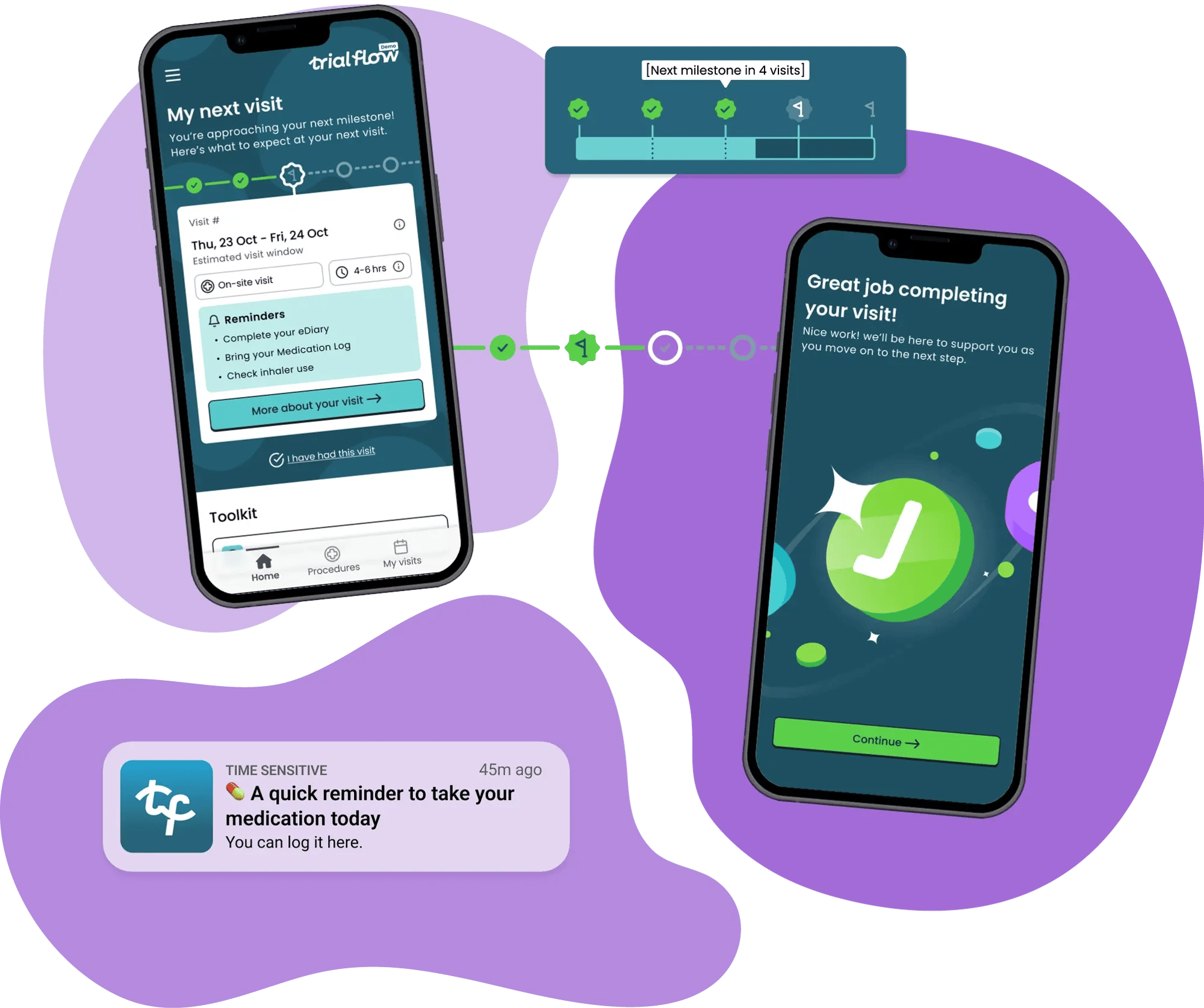
Motivation & Progress
Trial Flow's motivation & progress support Trial Flow uses behavioural science to help patients recognise how far they’ve come and understand the value of their participation.
Feature examples:
- Notifications
- Medication reminders
- Progress milestones
Reinforces progress and purpose to help patients stay motivated, adhere to requirements, and remain engaged through long or complex studies.
Support that fits the patient, the protocol and the trial journey
The same patient support strategy will not work for every protocol or patient population. Trial Flow provides one platform to deliver tailored support across ages, user types and studies.
Protocol-aligned
Support reflects the study schedule, procedures, visit requirements and key participation moments.
User-specific
Content and experience are adapted for adult, adolescent, or paediatric patients and their caregivers.
Journey-based
Support can adapt across visits, study stages, arms and sub-studies.
Portfolio-ready
Reusable support components enable this patient-centric experience to scale across research portfolios.

We are proud to have supported
50,000+
patients & caregivers
17+
countries worldwide
9
top global pharma & CROs
Background
Trial Flow supported a complex global study with a high-burden protocol, helping reduce patient dropout, accelerate projected timelines and improve site engagement.
Study details: 68 patients enrolled | 35 sites | 9 countries | 16 languages
Key proof points
- 53% reduction in dropout
- 14 months faster study delivery
- 83% site uptake
- 100% sponsor, CRO and site satisfaction
Turn patient support into actionable insight

Trial Flow captures and structures patient interaction data across each study, helping teams understand what patients use, value and ignore.
As this data builds across studies and therapy areas, it creates a clearer picture of where friction starts, when support is needed most, and what improves engagement, adherence and retention.
- See how patients engage with different types of support
- Identify confusion or disengagement earlier
- Inform retention strategies across patient cohorts and therapy areas
As your Trial Flow dataset grows, we help you build a stronger evidence base for improving patient support across your full research portfolio.




_screen%20use.png)

.png)
.png?width=1500&height=1500&name=Trial%20Flow%20Phone%20(Left).png)