Case study
Reducing dropout by 53% and cutting study timelines by 14 months in a global gastroenterology study

Key Results
53%
reduction in dropout rate
14 month
faster study delivery
100%
sponsor, CRO & site satisfaction
200+
users actively engaged
Background
Through our Trial Flow platform, Little Journey partnered with a global pharmaceutical sponsor and leading CRO to support a challenging gastroenterology study in erosive esophagitis.
Study details: 68 patients enrolled | 35 sites | 9 countries | 16 languages
Challenges
Burdensome protocol
The study protocol included multiple endoscopies under general anaesthetic for an already approved drug, placing significant burden on patients and families. Maintaining motivation to continue participation was identified as a key operational risk.
High expected dropout rate
As a result of this burden, the anticipated dropout rate was high (75%), posing a significant risk to study timelines and data completeness.The sponsor required a scalable patient support solution to reduce dropout without increasing burden on site teams.
Our solution
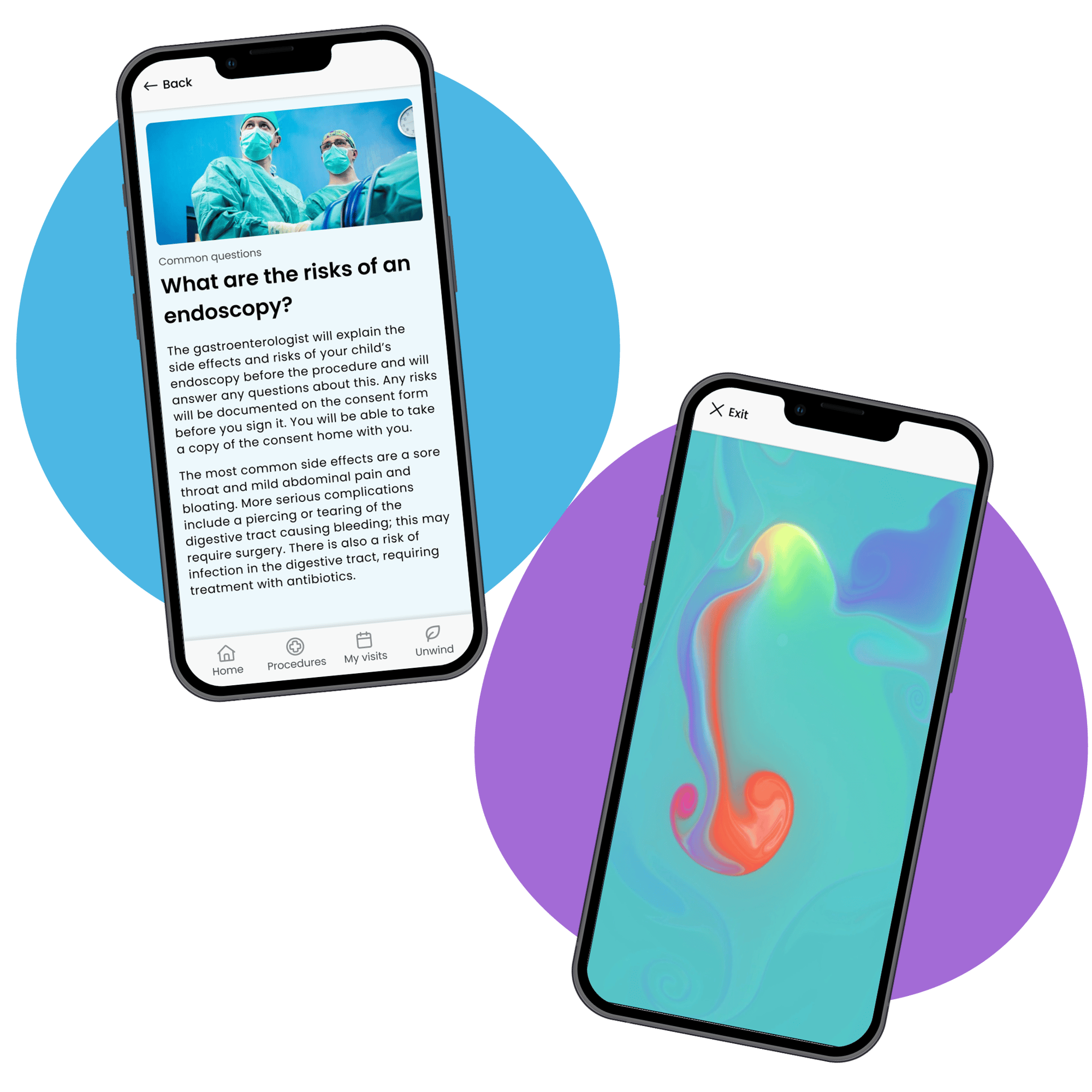
Trial Flow delivered a study-branded patient and caregiver support app focused on preparing families for procedures and guiding them through each visit.
Key elements of the app configured for this protocol included:
- Targeted, user-appropriate education to prepare patients and caregivers for endoscopies and anesthesia
- Clear, structured guidance on what to expect at each stage of the study
- Engaging content, including virtual tours, caregiver information, and distraction activities
Outcomes
Improved retention
53% reduction in dropout rate
75% anticipated → 35% actual
Faster delivery
14 month timeline reduction
October 2027 planned → August 2026 projected
High uptake
83% site uptake
29/35 sites, with 200+ cumulative app users
High satisfaction
100% would use again
With 75% of sites reporting it was easy to incorporate
The visits to the sites by the Trial Flow team were very valuable in helping the sites engage with the service and appreciate the value of implementing it.
Sponsor feedback
[Overview of the outcomes. Rearrange the cards below as needed!]
You may also like...
Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]

Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]

Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]
