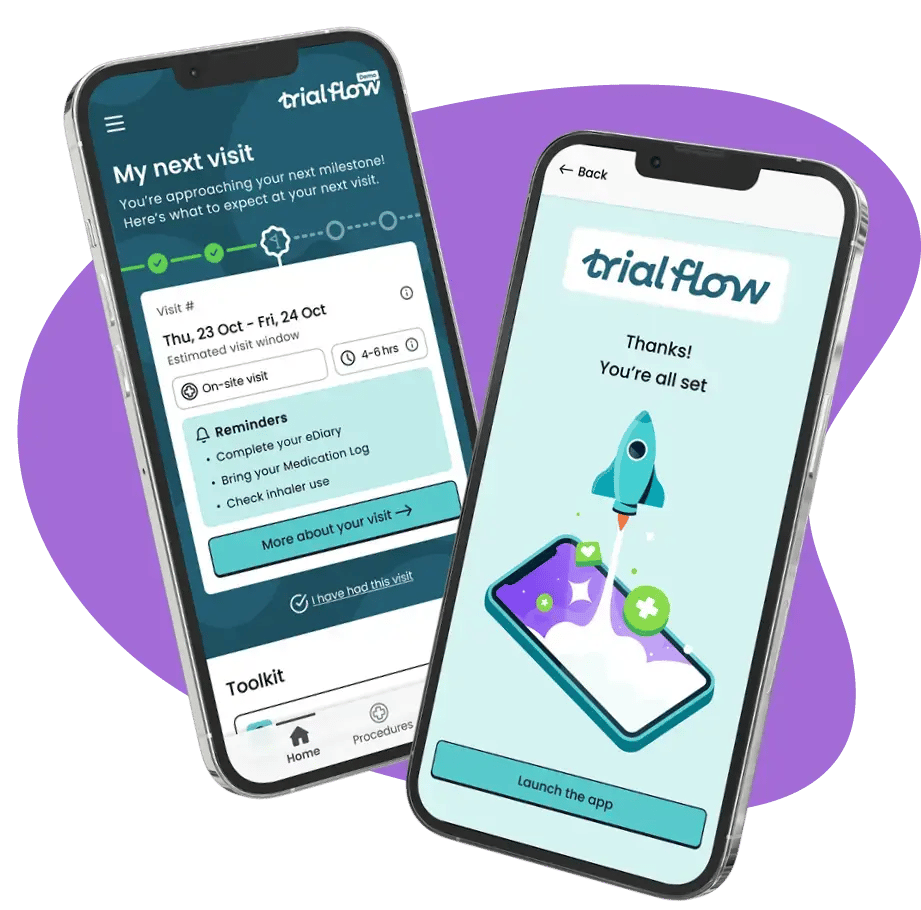
Case study
Streamlining site activation for a global multi-site trial
.webp?width=884&height=666&name=Trial%20Flow_Site%20Activation%20Case%20Study_Visual%201%20(1).webp)
Key Results
8 sites activated
across five countries, in one day
Under 60 minutes
training time per site
9/10 promoter score
very likely to recommend Trial Flow
100% satisfaction
with the effectiveness of training and activation
Background
Trial Flow activated global research sites quickly and consistently, enabling site teams to introduce the patient support delivery platform confidently without adding unnecessary operational burden. Patients could then be introduced to Trial Flow to support their trial experience, adherence and retention rates.
Challenge
Meeting trial timelines
80% of clinical trials are delayed. Multi-site trials are more likely to experience delays because of increased coordination, complexity and variation across sites.
Burden on site staff
In this study, research staff were managing multiple trials with different processes and tools. The sponsor needed a patient support solution that could integrate smoothly into existing workflows and be introduced confidently by site teams.
Our solution
A structured activation model for research sites
Trial Flow was implemented using a standardised site activation process designed to support rapid, consistent rollout.
This included:
- Standardised onboarding process across all sites
- Combination of remote and on-site activation sessions
- Training, demo, and onboarding delivered in under 60 minutes per site
- Support led by site success managers with clinical research experience
The approach helped sites understand the value of Trial Flow and feel confident introducing it to patients and caregivers.

Outcomes
Fast activation
8 sites activated in one day
Trial Flow was launched across five countries in a single day.
High satisfaction
9/10 likelihood to recommend
Sites were very likely to recommend Trial Flow to a colleague.
High uptake
95% average site uptake
Across all trials, Trial Flow has maintained strong average site uptake.
Strong site confidence
100% training satisfaction
Site teams were satisfied with the effectiveness of training and activation.
I knew of the app benefits, but seeing it first-hand really gave me much better insight to how easy, useful, beneficial, and user friendly it really is.
Clinical Research Associate, US Site
[Overview of the outcomes. Rearrange the cards below as needed!]
You may also like...
Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]

Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]

Case study
[Case study title]
[Summary of the case study, preferably a simplified version of the background or problem]